Sponsored Programs Basics
Quick Reference Guide
Introduction
Sponsored programs – when a UMB employee wants to conduct research, run clinical trials, or complete other projects, programs, or services that require funding, they can do so through a sponsored program and receive funding from an external organization.
Main players – The principal investigator (PI) is the individual who oversees the sponsored project. The project team includes the PI and their collaborators both within UMB, like other faculty members and postdoctoral fellows, and outside of UMB that work for other organizations. The sponsor is the organization funding the program external to UMB like NIH, NSF, other federal agencies, corporations, or nonprofits. The recipient is the organization that receives the award; this would be UMB.
Research Administration
Research administration supports the project team by ensuring regulations are followed.
SPA (Sponsored Programs Administration) oversees proposal routing and submission, issues subawards, approves non-financial post-award transactions (changes to the project and budget).
CCT (Center for Clinical Trials and Corporate Contracts) supports projects that have for-profit sponsors and negotiates corporate-funded sponsored project agreements.
SPAC (Sponsored Project Accounting and Compliance) oversees the financial system and sponsor payments and manages audits.
Regulatory Offices
Human Research Protections is a comprehensive program that affords protections for all human research participants.
Veterinary Resources supports the use of animal models of human biology in translational biomedical research and discovery.
Office of Animal Welfare Assurance is the regulatory oversight office for the UMB Animal Care and Use Program and provides support for the UMB Institutional Animal Care and Use Committee (IACUC).
Environmental Health and Safety (EHS) supports UMB's research enterprise by protecting laboratory workers, the environment, and the community from potential exposure to hazardous materials while also protecting the integrity of experimental materials.
Conflict of Interest (COI) is the University’s designated office for the management of financial conflicts of interest in research. As UMB employees, disclosure of certain financial interests are required before engaging in certain professional activities, in compliance with the State of Maryland Public Ethics Law, U.S. Public Health Service Federal Regulations and Campus Policies.
Grant Lifecycle
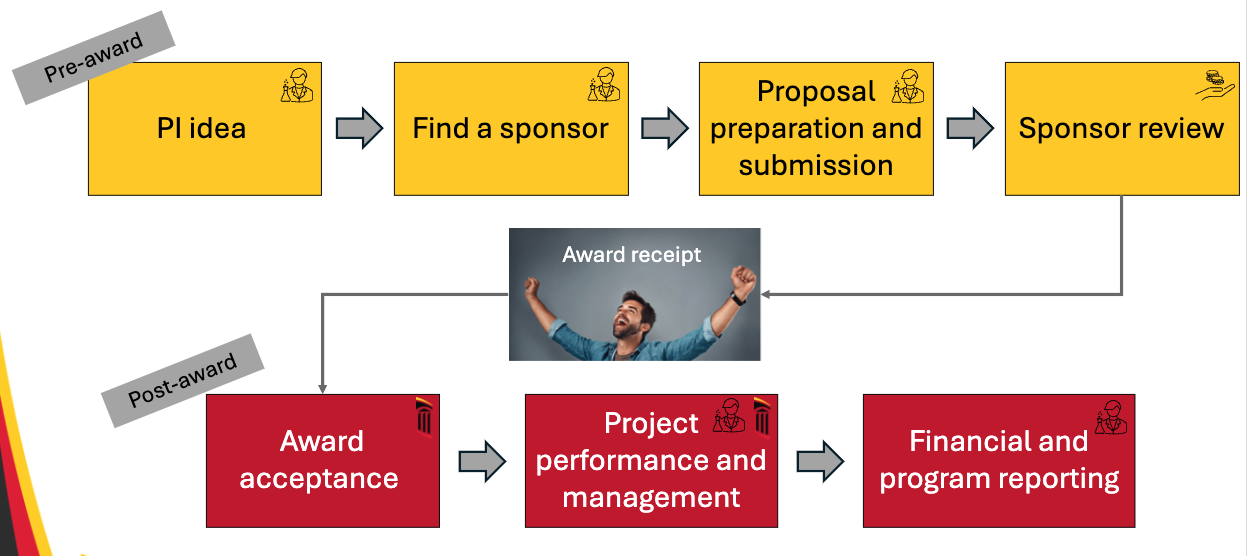
- The PI has an idea for a project related to research, training, or service.
- The PI needs to find a sponsor to fund the project.
- The PI, with the help of a department administrator, drafts a proposal with a description, research plan, and budget for the award and subawards based on the sponsor’s requirements. The proposal is entered into Kuali Research, approved by the department, the dean, and UMB’s central office, and then submitted.
- The sponsor reviews the proposal submitted by the PI and makes the decision to fund it or not. If the proposal is rejected, the sponsor will often provide review notes to provide reasoning for the rejection and guide resubmission.
- If the sponsor agrees to funding, the award will be issued to UMB. The award will be an agreement that describes the project, funding, terms and conditions, dates, report requirements, requested deliverables, and penalties. There are different kinds of awards including grants, contracts and cooperative agreements.
- UMB reviews the terms of the award, accepts it, and sets up a project account.
- The project begins, managed by the PI and unit research administrators.
- The PI is responsible for reporting, including how the funding is being used and providing progress reports at the end of every budget period, usually one year long.


